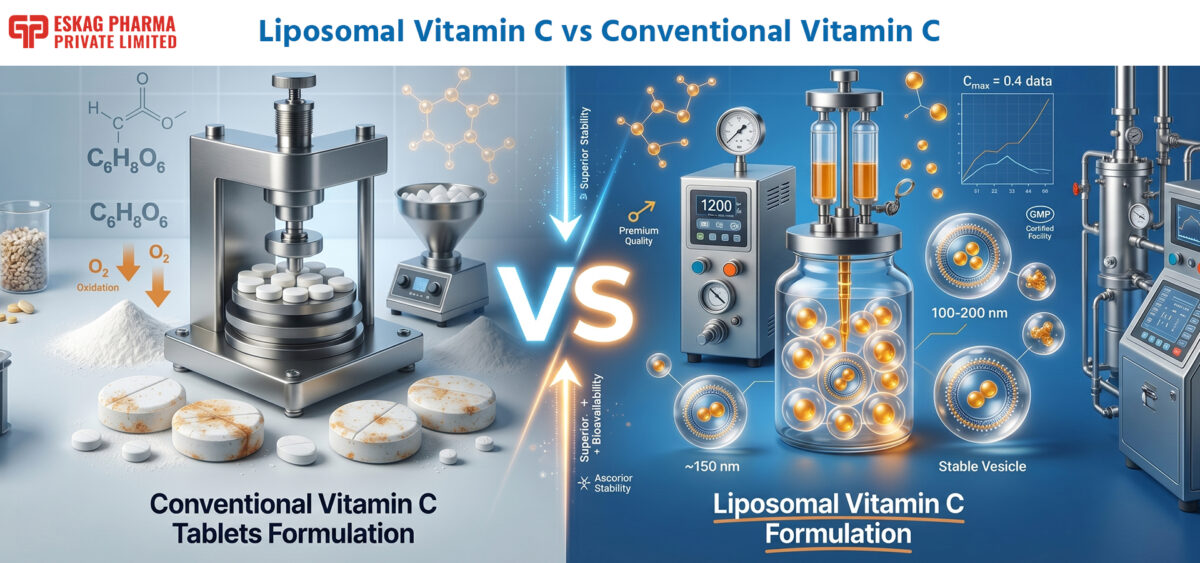
Liposomal Vitamin C vs Conventional Vitamin C: A Complete Manufacturing Perspective
Key Takeaways
- Evolution of Delivery: The traditional manufacturing process of vitamin c is being replaced by liposomal technology to dramatically increase cellular bioavailability.
- Formulation Challenges: A stable liposomal vitamin c formulation requires advanced cold-process techniques to protect the active ingredient from oxidation, unlike standard solid dosages.
- Market Advantage: Brands highlighting liposomal vitamin c benefits can command premium pricing due to superior gastric comfort and better absorption rates.
- Choosing the Right Partner: Partnering with an expert liposomal vitamin c manufacturer like Eskag Pharma ensures consistent vesicle size, stability, and WHO-GMP compliance.
In the rapidly evolving nutraceutical industry, the choice between liposomal vitamin C and conventional vitamin C goes far beyond simple formulation differences—it shapes everything from production scalability to end-user efficacy. As a leading liposomal vitamin C manufacturer, Eskag Pharma brings a unique manufacturing lens to this comparison. With advanced encapsulation technologies and decades of expertise in third-party nutraceutical production, we understand the technical, regulatory, and commercial realities that brands face when choosing between traditional vitamin C tablets formulation and cutting-edge liposomal vitamin C formulation.
This manufacturing perspective explores the evolution, processes, complexities, and real-world advantages, helping healthcare brands and supplement manufacturers make informed decisions. Whether you are sourcing from a trusted liposomal vitamin C supplier or evaluating your own production capabilities, the shift toward liposomal systems represents a significant leap in how vitamin C is delivered to consumers.
The Evolution of Vitamin C in Nutraceutical Manufacturing
Vitamin C, or L-ascorbic acid, has long been a cornerstone of immune support, antioxidant defense, and collagen synthesis. However, the journey from raw active ingredient to finished supplement has transformed dramatically over the past few decades.
The Traditional Manufacturing Process of Vitamin C
The manufacturing process of vitamin C for commercial use primarily follows two established routes. The classic Reichstein process is a combined chemical and microbial method that starts with D-glucose, involving hydrogenation to D-sorbitol, fermentation to L-sorbose, protection steps, chemical oxidation to 2-keto-L-gulonic acid, and final conversion to L-ascorbic acid. In contrast, the modern two-step fermentation process—widely adopted especially in large-scale production—uses microbial fermentation for both key conversion steps (D-sorbitol to L-sorbose, followed by L-sorbose to 2-keto-L-gulonic acid), offering advantages in lower energy use, reduced hazardous solvents, and overall cost efficiency while maintaining high product quality.
For conventional products, the next stage is straightforward: the ascorbic acid is blended with excipients such as binders, disintegrants, and lubricants before compression into tablets or capsules. This vitamin c tablets formulation approach relies on high-speed mixers, granulators, and tablet presses—equipment that has been standardized across the pharmaceutical and nutraceutical industries for decades. The entire process is cost-effective, scalable, and requires minimal specialized infrastructure.
Limitations of a Standard Vitamin C Tablets Formulation
Despite its simplicity, the vitamin c tablets formulation has inherent constraints. Ascorbic acid is highly water-soluble and prone to oxidation when exposed to heat, light, moisture, or oxygen. In tablet form, it dissolves rapidly in the stomach, leading to saturation of intestinal transporters (SVCT1 and SVCT2), which limits absorption at higher doses. Excess unabsorbed vitamin C can cause gastric distress, including bloating, diarrhea, and discomfort—especially problematic for high-dose supplementation.
Bioavailability studies consistently show that conventional forms achieve only partial cellular uptake, with much of the dose excreted unused. These limitations have driven the industry shift toward advanced delivery systems that protect the active ingredient and enhance absorption.
The Industry Shift Towards Advanced Liposomal Delivery Systems
Today’s forward-thinking liposomal vitamin c manufacturer recognizes that consumer demand for higher efficacy is reshaping production priorities. The manufacturing process of vitamin c now extends beyond bulk synthesis into sophisticated encapsulation technologies. Brands are moving away from basic vitamin c tablets formulation toward liposomal vitamin C formulation because it addresses the core shortcomings of traditional methods while opening new market opportunities in premium wellness segments.
Understanding the Liposomal Vitamin C Formulation Process
Liposomal technology represents a paradigm shift in nutraceutical science, using nature-inspired delivery vehicles to protect and deliver vitamin C more effectively.
The Science of Phospholipids and Spherical Vesicles
At the heart of liposomal vitamin C formulation lies the phospholipid bilayer—typically derived from non-GMO sunflower or soy lecithin. These amphiphilic molecules self-assemble into spherical vesicles (liposomes) with a hydrophilic aqueous core and a hydrophobic lipid shell. L-ascorbic acid is encapsulated within the aqueous core, shielded from external degradative factors.
The resulting nano-sized particles (typically 100–200 nm) mimic human cell membranes, enabling enhanced uptake. Proposed mechanisms include membrane fusion and endocytosis, which facilitate better delivery compared to standard transporter-limited absorption. This structural innovation is what sets a professional liposomal vitamin c manufacturer apart from conventional producers.
Overcoming Oxidation in a Liquid Liposomal Vitamin C Formulation
Vitamin C’s greatest enemy is oxidation. In a liquid liposomal vitamin c formulation, manufacturers must employ advanced techniques to maintain stability. Cold-process methods, nitrogen purging, and oxygen-free environments prevent the degradation of the enediol group in L-ascorbic acid. High encapsulation efficiency (EE%)—often exceeding 70–80%—ensures the majority of the active remains protected inside the vesicle rather than exposed on the surface.
Cold-Process Techniques to Preserve Active L-Ascorbic Acid
Unlike heat-intensive tablet production, liposomal vitamin c formulation relies on cold-process techniques such as high-pressure homogenization or microfluidization. These methods create uniform nano-emulsions without compromising the heat-sensitive ascorbic acid. Sonication and controlled-pressure cycles further refine particle size and distribution, resulting in stable, ready-to-use liquid or powder formats that retain full potency throughout shelf life.
Contrasting the Manufacturing Complexities and Infrastructure
The leap from conventional to liposomal production demands entirely different infrastructure and expertise.
Equipment Needs: Standard Blenders vs. Microfluidizers
A standard vitamin c tablets formulation line uses basic ribbon blenders, fluid bed dryers, and rotary tablet presses—equipment found in most nutraceutical facilities. In contrast, a dedicated liposomal vitamin c manufacturer invests in specialized machinery: high-pressure homogenizers, microfluidizers, and liposome extruders. These systems require precise control of pressure, temperature, and shear forces to achieve consistent vesicle formation. The capital investment is higher, but it enables premium product differentiation.
Stability Testing: Why Liquid Liposomes are Harder to Master than Tablets
Stability testing for liposomal products is far more rigorous. Liquid formulations must withstand thermal stress, pH fluctuations, and long-term storage without leakage or vesicle fusion. Manufacturers monitor particle size distribution via dynamic light scattering, zeta potential for colloidal stability, and EE% over time. Tablets, by comparison, are inherently more stable due to their dry state, making conventional production simpler from a quality-control standpoint.
Cost-Benefit Analysis for a Premium Liposomal Vitamin C Supplier
While upfront costs are higher for a liposomal vitamin c supplier, the return on investment is compelling. Premium positioning allows higher pricing, stronger consumer loyalty, and repeat purchases driven by noticeable efficacy. Brands partnering with an experienced liposomal vitamin c manufacturer gain access to differentiated products that justify the added complexity.
Bridging the Gap: Translating Science into Consumer Demand
The true value of liposomal technology emerges when science meets real consumer needs.
Why the Best Liposomal Vitamin C Benefits Outweigh Standard Pills
Clinical studies demonstrate that liposomal vitamin c benefits include improved bioavailability, with many human trials reporting approximately 1.5–2.4 times higher absorption (measured by AUC and Cmax) compared to non-liposomal forms, depending on the specific formulation and study conditions. Enhanced plasma and leukocyte levels translate to superior immune support, antioxidant protection, and collagen synthesis—benefits consumers can actually feel.
Preventing Gastric Distress in High-Dose Supplementation
One of the most appreciated liposomal vitamin c benefits is reduced gastrointestinal side effects. By protecting vitamin C until it reaches the absorption site, liposomal delivery minimizes direct contact with stomach lining, allowing higher doses without the discomfort common in standard tablets.
How Healthcare Brands are Upgrading Their Product Portfolios
Progressive brands are actively replacing or supplementing traditional lines with liposomal options. A reliable liposomal vitamin c manufacturer enables rapid portfolio upgrades, helping companies capture the growing premium supplement segment driven by informed consumers seeking maximum efficacy.
Quality Control and Regulatory Compliance in Encapsulation
Excellence in manufacturing is defined by uncompromising quality systems.
Measuring Particle Size and Encapsulation Efficiency (EE%)
Advanced liposomal vitamin c manufacturer facilities employ laser diffraction and HPLC methods to verify particle size (<200 nm) and EE% (>70%). These metrics directly correlate with product performance and batch consistency.
FSSAI and WHO-GMP Standards for Liposomal Supplements
Compliance with FSSAI and WHO-GMP guidelines is non-negotiable. Stringent documentation, validated processes, and regular audits ensure every batch meets international safety and efficacy standards—critical for both domestic and export markets.
Maintaining Batch-to-Batch Consistency as a Liposomal Vitamin C Manufacturer
As a liposomal vitamin c manufacturer, maintaining consistency requires tight process controls, real-time monitoring, and robust SOPs. Eskag Pharma’s state-of-the-art facility in Haridwar exemplifies this commitment, delivering uniform quality across every production run.
Why Choose Eskag Pharma as Your Manufacturing Partner?
When selecting a manufacturing partner for advanced nutraceuticals, experience, infrastructure, and end-to-end capability matter most.
India’s Trusted Liposomal Vitamin C Manufacturer with 47+ Years Legacy
Eskag Pharma stands as India’s trusted liposomal vitamin c manufacturer with over 47 years of legacy in pharmaceutical and nutraceutical excellence. Our WHO-GMP certified facility in Haridwar combines cutting-edge technology with time-tested quality systems.
End-to-End Support from Liposomal Vitamin C Formulation to Packaging
We provide complete support—from custom liposomal vitamin c formulation development to regulatory documentation and innovative packaging solutions. Whether you need liquid shots, capsules, or powders, our team handles every stage with precision.
Your Reliable Export-Ready Liposomal Vitamin C Supplier
As a reliable liposomal vitamin c supplier, Eskag Pharma serves global markets with export-ready products that meet stringent international standards. Our liposomal vitamin c formulation expertise, combined with proven liposomal vitamin c benefits, positions your brand for success in competitive wellness markets.
Partner with Eskag Pharma—the liposomal vitamin c manufacturer that transforms scientific innovation into market-leading supplements. Contact us today to discuss your next product launch and experience the difference that true manufacturing expertise delivers.
-
 📅 May 28, 2026
📅 May 28, 2026
Anaemia is one of the most common and consequentia... -
 📅 May 27, 2026
📅 May 27, 2026
Patients struggling with iron deficiency anaemia o... -
 📅 May 27, 2026
📅 May 27, 2026
Patients struggling with iron deficiency anaemia o... -
 📅 May 26, 2026
📅 May 26, 2026
Patients with iron deficiency anaemia often strugg... -
 📅 May 22, 2026
📅 May 22, 2026
Curcumin, the golden active compound in turmeric, ... -
 📅 May 20, 2026
📅 May 20, 2026
Curcumin, the active compound in turmeric, is cele... -
 📅 May 19, 2026
📅 May 19, 2026
Curcumin, the active compound in turmeric, is wide... -
 📅 May 18, 2026
📅 May 18, 2026
Sleep is essential, yet millions struggle to achie... -
 📅 May 18, 2026
📅 May 18, 2026
Women’s health supplements form one of the m... -
 📅 May 18, 2026
📅 May 18, 2026
Every supplement brand that sells a probiotic prod...
Conventional vitamin C is typically delivered as free L-ascorbic acid in tablets or capsules (vitamin c tablets formulation). Liposomal vitamin C encapsulates the active ingredient inside phospholipid vesicles, protecting it from degradation and enabling enhanced absorption through different uptake mechanisms.
The initial manufacturing process of vitamin c (Reichstein or two-step fermentation) remains the same to produce bulk ascorbic acid. The key difference lies in the downstream liposomal vitamin c formulation stage, which requires high-shear homogenization, microfluidization, and cold-processing techniques instead of simple blending and compression used in standard tablet production.
Liposomal vitamin c benefits include significantly better bioavailability (often 1.5–2.4 times higher plasma levels), reduced gastric distress at high doses, prolonged blood levels, and improved cellular uptake into immune cells—resulting in stronger antioxidant and immune support compared to standard pills.
Standard vitamin c tablets formulation relies on saturable intestinal transporters (SVCT1/SVCT2). Once these are saturated, excess vitamin C passes unabsorbed, leading to lower overall uptake and potential gastrointestinal side effects. Rapid dissolution in the stomach also exposes the molecule to oxidative degradation.
A dedicated liposomal vitamin c manufacturer requires high-pressure homogenizers, microfluidizers, liposome extruders, and precise particle-sizing instruments—far more advanced than the standard blenders, granulators, and tablet presses used for conventional vitamin c tablets formulation.
Stabilization involves cold-process manufacturing, nitrogen purging to remove oxygen, high encapsulation efficiency (>70–80%), addition of natural stabilizers like gum arabic or cholesterol, and strict control of pH, temperature, and light exposure during filling and storage to prevent oxidation and vesicle leakage.
Brands should seek a liposomal vitamin c supplier with proven WHO-GMP certification, documented encapsulation efficiency and particle size data, batch-to-batch consistency records, end-to-end formulation-to-packaging capability, and successful export experience to ensure regulatory compliance and product performance.
Yes, high-quality liposomal encapsulation significantly reduces oxidation by shielding L-ascorbic acid inside the aqueous core of the phospholipid vesicle, away from oxygen, light, and metal ions. Combined with cold processing and inert gas protection, it markedly improves shelf-life stability compared to free ascorbic acid.
Absolutely. As an established liposomal vitamin c manufacturer with a WHO-GMP certified facility in Haridwar and over 47 years of legacy, Eskag Pharma offers scalable production volumes, customized liposomal vitamin c formulation, and full export documentation support for markets across Africa, LATAM, Southeast Asia, Middle East, and beyond.
Liposomal vitamin c manufacturer facilities use advanced analytical tools such as dynamic light scattering (DLS), laser diffraction, and transmission electron microscopy (TEM) to measure particle size distribution (typically targeting 100–200 nm) and confirm uniformity, which directly impacts stability and bioavailability.
